  
It has SI units of joules per kelvin (JK 1) or kgm 2 s 2 K 1. Hence a macroscopic sample of a gas occupies all of the space available to it, simply because this is the most probable arrangement.Įntropy depends not only on the number of atoms or molecules and the volume of available space, but also their freedom of motion, which corresponds to temperature and state of matter. In equations, the symbol for entropy is the letter S. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system. In this manner, the quantity S is utilized as a type of internal ordering energy, which accounts. In chemistry, entropy units can be utilised to represent enthalpy changes. Entropies of gases are much larger than those of condensed phases. Unit of Entropy: The entropy unit seems to be a non-SI measurement for thermodynamic entropy that seems to be equivalent to one calorie per kelvin per mole, or approximately 4.184 joules per kelvin per mole. The entropy also increases as the pressure or concentration becomes smaller. The probability of arrangements with essentially equal numbers of molecules in each bulb is quite high, however, because there are many equivalent microstates in which the molecules are distributed equally. entropy, the measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work. The SI unit of entropy is joule per kelvin (JK1). The entropy of a substance increases with its molecular weight and complexity and with temperature. Although nothing prevents the molecules in the gas sample from occupying only one of the two bulbs, that particular arrangement is so improbable that it is never actually observed. Entropy (S) has two definitions: Entropy is a measure of disorder in a system. Weve successfully rearranged the equations Entropy - Key takeaways. Therefore: TS° total is a version of Gibbs free energy. Instead of four molecules of gas, what if we had one mole of gas, or 6.022 × 10 23 molecules in the two-bulb apparatus? If we allow the sample of gas to expand spontaneously in the two containers, the probability of finding all 6.022 × 10 23 molecules in one container and none in the other at any given time is extremely small, effectively zero. The units of entropy are J K-1 mol-1, whilst the units of Gibbs free energy are kJ mol-1. It equals to the total entropy (S) divided by the total mass (m). 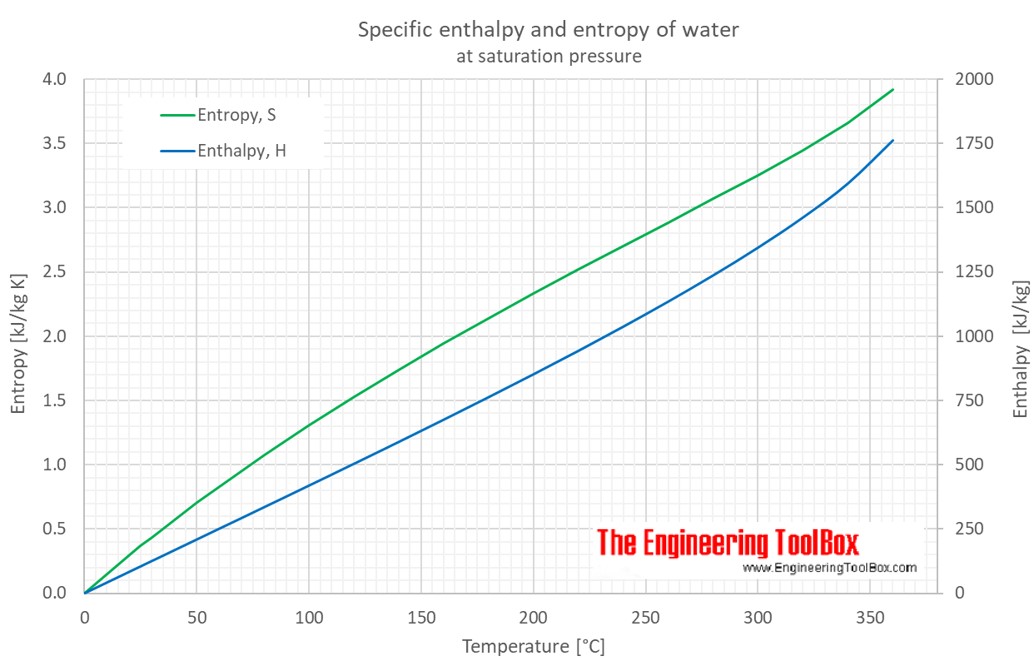
which reduces the system performance.\( \newcommand\): The Possible Microstates for a Sample of Four Gas Molecules in Two Bulbs of Equal Volume The specific entropy (s) of a substance is its entropy per unit mass. In the same article in which he introduced the name entropy, Clausius gives the expression for the entropy production for a cyclical process in a closed system, which he denotes by N, in equation (71) which reads Information entropy is a measure of the 'spread. Thermodynamic entropy bears a close relationship to the concept of information entropy (H). Statistical mechanics and information entropy. In 1865 Rudolf Clausius expanded his previous work from 1854 on the concept of "unkompensierte Verwandlungen" (uncompensated transformations), which, in our modern nomenclature, would be called the entropy production. The entropy at another temperature is then equal to the increase in entropy on heating the system reversibly from absolute zero to the temperature of interest. J/K) used for entropy in the thermodynamic definition follow from a historical association with heat transfer under temperature gradients, in other words, the definitions of temperature and entropy are intertwined, with entropy being the more fundamental property. Consider a system consisting of two objects, each containing two particles, and two units of thermal energy (represented as ) in Figure 16.9. The importance of avoiding irreversible processes (hence reducing the entropy production) was recognized as early as 1824 by Carnot. The units of of energy over temperature (e.g. The number of available microstates increases when matter becomes more dispersed, such as when a liquid changes into a gas or when a gas is expanded at constant temperature. Development of entropy in a thermodynamic systemĮntropy production (or generation) is the amount of entropy which is produced during heat process to evaluate the efficiency of the process.Įntropy is produced in irreversible processes. ENE4.A.1 (EK), ENE4.A.2 (EK) Google Classroom About Transcript According to the Boltzmann equation, entropy is a measure of the number of microstates available to a system. Since entropy also depends on the quantity of the substance, the unit of entropy is calories per degree per mole (or) eu. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
